The gaps in medicine quality data
The use of substandard and falsified medicines threatens the lives of many millions of people across the world. Although this issue is common in many regions where malaria is endemic, our understanding of the full extent of the problem and its consequences is remarkably limited.
A team of researchers from the Worldwide Antimalarial Resistance Network (WWARN) has worked for the last 2 years to build a comprehensive open-access global database that collates and analyses all reports on antimalarial medicine quality going as far back as 1946, in English, French and Spanish. The studies provide a valuable oversight of the scale and severity of the issue of falsified (due to fraud) and substandard medicines (due to laboratory errors or storage of medicines), and highlight the tremendous gap in available data on the quality of medicines used in treatment of malaria patients.
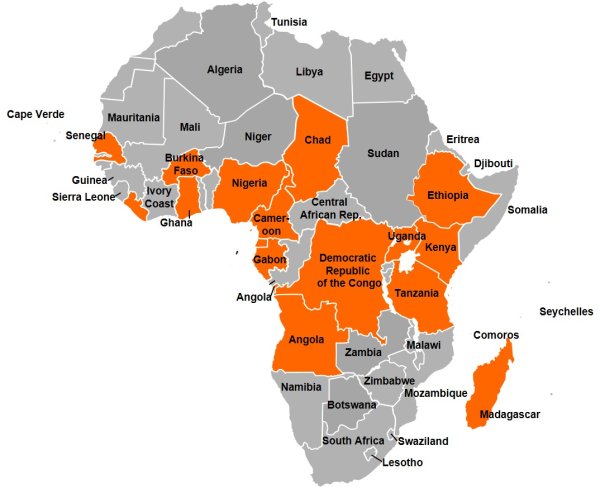
There are no publically available reports on the quality of malaria treatments from over 60 per cent of malaria endemic countries. In Africa alone, an estimated 200-400 million courses of antimalarial treatments are administered each year, with an additional 100 million courses elsewhere. In spite of this figure, data on drug quality has only been analysed and presented publicly for ~9,000 drug samples over the past 60 years.
Countries where publicly available antimalarial drug quality reports have been found over the last 5 years.
“Estimates of medicine quality vary greatly depending on the sampling methodology and the technique used, but our limited knowledge of the full extent of the problem renders us incapable of adequately assessing how to respond” says Patricia Tabernero, first author of a recent paper in Malaria Journal and the Scientific Coordinator of Drug Quality at WWARN.
The team found that of information gathered on 9,348 antimalarial medicine samples, 30 per cent failed either chemical or packaging quality tests. Of the drugs that failed tests, more than 39 per cent were classified as being falsified, 2.3 per cent as substandard and more than 58 per cent were classified as of ‘poor quality’ without evidence available to categorize them as either substandard or falsified.
“It is imperative that we invest in the improvement of sampling protocols and techniques within the areas where malaria is endemic. There needs to be the investment to build the capacity of the quality control laboratories so we can obtain reliable test results and so we can be better equipped to face this growing problem,“ says Souly Phanouvong PharmD, Ph.D., Manager for Asia with the Promoting the Quality of Medicines Program (PQM).1
Souly goes on to suggest, “In order to respond, we need to work in partnership with governments, health care professionals, the private sector, research institutes, the donors and NGOs to determine the true scale of the problem, and empower Medicine Regulatory Authorities with realistic strategies and enforcement capabilities to improve the treatment of millions of patients.”
Poor medicine quality is not just restricted to antimalarial treatments, but is a hugely underrated universal global health burden. One key cause for this information gap is that laboratories in the regions most affected lack the resources needed to analyse drug samples and screen suspect medicines.
“More than 600,000 people die from malaria infection each year – many of these cases could be avoided if drugs available to patients were efficacious, high quality, and used correctly. In addition, poor quality antimalarials, especially those that are substandard are likely to be important drivers of drug resistance” says Dr Paul Newton, senior author of the study and Director of the Lao-Oxford-Mahosot Hospital-Wellcome Trust Research Unit in Laos. “We need to ensure that assay techniques and standardised sampling methods to define different types of poor drug quality are accessible in countries where malaria is endemic, and we need to place this issue higher up the global public health agenda.”
Download the latest article: Tabernero, P., et al., Mind the gaps - the epidemiology of poor-quality antimalarials in the malarious world - analysis of the Worldwide Antimalarial Resistance Network database. Malaria Journal, 2014; 13:139; doi:10.1186/1475-2875-13-139
NEWS FLASH: Lao-Oxford-Mahosot Hospital-Wellcome Trust Research Unit & WWARN have recently confirmed an agreement to provide advice and assistance on medicine quality issues to the Joint Inter-Agency Task Force (JIATF) Project, focusing on the theft, diversion, and counterfeiting of Global Fund and USAID-funded anti-malarial medicines. The JIATF comprises personnel from The Global Fund's Office of Inspector General (OIG), USAID's Office of the Inspector General (USAID-OIG), and UNDPs Office of Audit and Investigations (OAI).
Other related publications:
- WHO Drug Alert No. 131, Falsified antimalarial medicines in west and central Africa, March 25, 2014
- Financial Times Special Report on Combatting Malaria, article entitled Call for Greater Penalties for Peddlers of Fake Antimalarials (published online April 25, 2014; restricted access may require login to FT.com)
- Hetzel MW et al. Quality of Antimalarial Drugs and Antibiotics in Papua New Guinea: A Survey of the Health Facility Supply Chain. PLoS ONE 9(5): e96810 doi:10.1371/journal.pone.0096810
- SciDev.net, Almost a third of malaria drugs failed quality tests (published April 25, 2014 online)
Footnote: 1 QM is a global health program funded by the U.S. Agency for International Development (USAID) and implemented by the U.S. Pharmacopeial Convention (USP)
